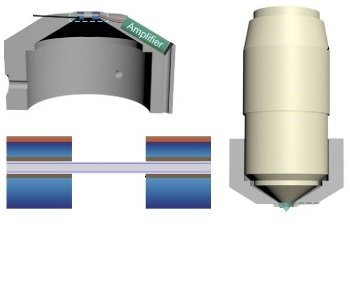
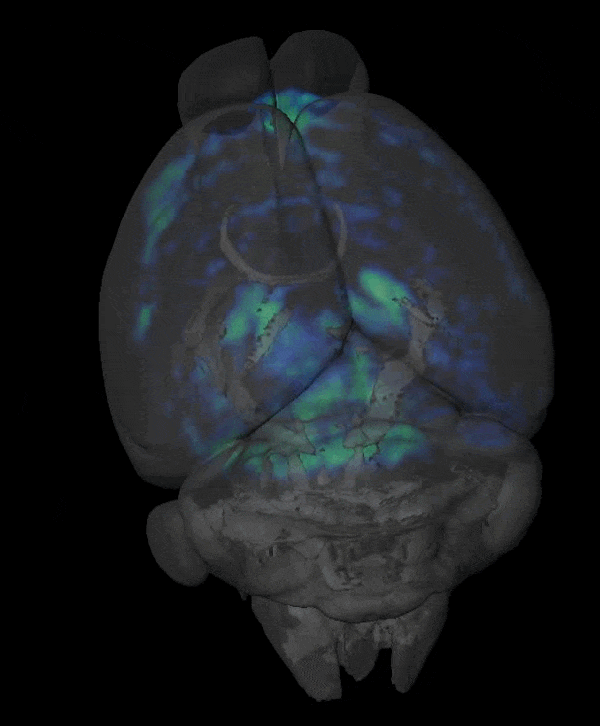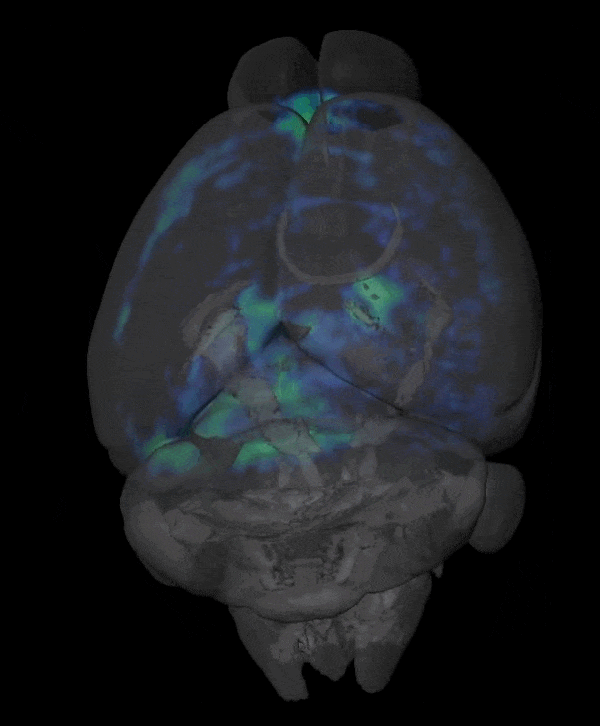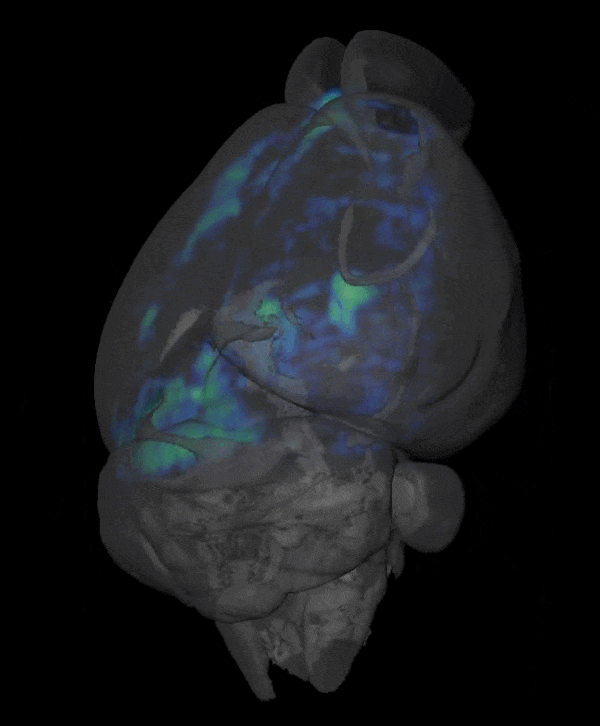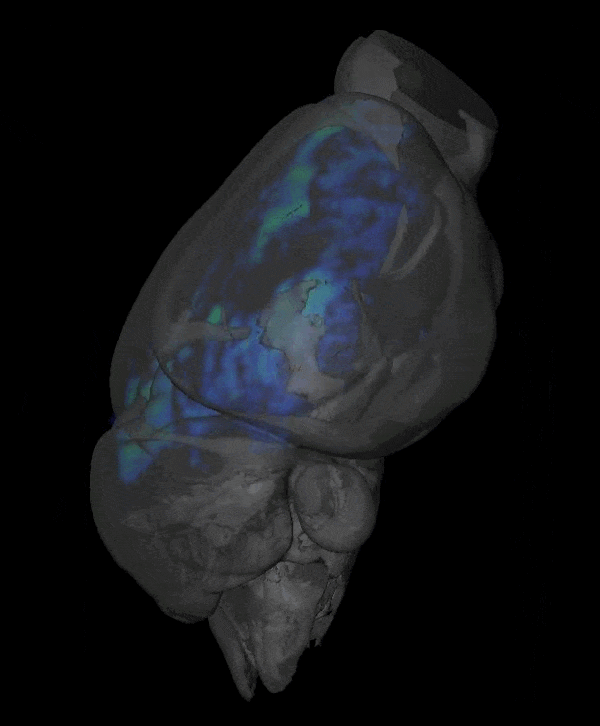
Novel Functional and Molecular Imaging Methods with Clinical Translation Potential
We work at the interface of engineering, physics, biology and medicine to devise novel tools for high performance functional and molecular imaging. The Research Centre is fully affiliated with the Faculty of Medicine, University of Zurich and the Department of Information Technologies and Electrical Engineering, ETH Zurich. It is also an integral part of the Institute for Biomedical Engineering (IBT) and the Institute of Pharmacology and Toxicology (IPT). Our research is focused on methods that can broadly impact pre-clinical research and clinical practice by delivering information presently not attainable with existing state-of-the-art imaging modalities. We contribute to the creation of these new technologies in several diverse ways, from the establishment of solid theoretical background, imaging instrumentation, inverse and machine learning methods, to the development of in vivo imaging methodologies and contrast enhancement approaches. We are also actively engaged in clinical trials involving the newly-developed imaging technology. Examples of projects include development of volumetric real-time tomography systems for pre-clinical molecular imaging, ultrafast microscopy for large-scale recording of deep brain activity, handheld clinical diagnostics systems.